last updated 11 November 2016
A number of collaborative research projects based around the Great Barrier Reef spawning period are active within the National Sea Simulator. These include:
- Dredging and coral reproduction
- Oil spills and coral spawning
- Assisted evolution
- Evolution in the 21st century: Can coral reef organisms adapt to climate change?
- Genetic markers of climate change adaptation in corals
- Studies of gene function using novel techniques
- Understanding crown-of-thorns starfish outbreaks
Dredging and coral reproduction
An Acropora millepora colony spawning. Photo: M. Nordborg
AIMS, Western Australian Marine Science Institute, University of Western Australia
Dr Andrew Negri
Dr Ross Jones
Student: Gerard Ricardo
and others
Sediments released from dredging activities can reduce or block light, clog feeding and smother coral. The sediments can also affect many aspects of coral reproduction and recruitment processes. This project aims to understand the impact of dredging sedimentation on early life history stages of coral.
Oil spills and coral spawning
Researcher prepares oil samples for ecotoxicology experiments.
AIMS, KAUST
Dr Andrew Negri
Dr Diane Brinkman
Dr Lone Hoj
Florita Flores
Student: Mikaela Nordborg
Prof. Carlos M. Duarte
Prof. Susana AgustÃ
Oil from spills and leaks can interact with corals at both the water's surface and beneath the ocean. In this study, millimetre-long coral will be exposed to oils under controlled conditions to measure the sensitivity of this critical stage in a coral's life history. We will be investigating the uptake of toxic oil components in corals via microalgae, as well as the effects of UV light on the toxicity of oils to corals. These results can be used by industry and regulators to assess the risks posed to corals by spills and leaks.
Assisted evolution
A researcher carefully collects coral eggs, preparing them for cross fertilisation with another species
AIMS, University of Melbourne, Hawaii Institute of Marine Biology, Swinburne University of Technology, ARC Centre of Excellence for Coral Reef Studies
Prof Madeleine van Oppen
Prof Ruth Gates
Mr Carlos Alvarez-Roa
Ms Lesa Peplow
Ms Wing Chan
Ms Katarina Damjanovic
Ms Leela Chakravarti
Prof Ary Hoffman
Prof Linda Blackall
Dr Mary Hagedorn
Dr Rebecca Hobbs
Dr Jon Daly
Mr Michael Henley
Prof Bette Willis
Dr Bill Leggat
This project will use 'assisted evolution' - the active genetic manipulation of native coral populations - to increase the rate of naturally occurring evolutionary processes in order to enhance certain traits such as environmental stress resistance and growth rate.
During this year's spawning event, two approaches will be followed:
1. Different coral species are crossed in order to create new genetic combinations on which selection can act to enhance stress tolerance. This component of the research has linkages to Paul Allen Foundation/Vulcan funding to Gates and van Oppen.
2. Sperm from a number of coral species and individuals will be cryo-preserved to expand the GBR coral cryo-repository held at Taronga’s CryoReserve in Taronga’s Western Plains Zoo in Dubbo (NSW). Further, AIMS staff and students will be trained in the cryo-preservation of cultured coral photosymbionts, Symbiodinium spp., and existing cultures at AIMS will be cryo-preserved. Early coral life stages will be (a) inoculated with frozen Symbiodinium spp and then thawed, and (b) inoculated with unfrozen Symbiodinium spp to assess whether the cryo-preserved symbionts are viable. This component of the work has financial support from the Great Barrier Reef Foundation.
Evolution in the 21st century: Can coral reef organisms adapt to climate change?
AIMS, Marsden Fund, Victoria University of Wellington
Dr Nicole Webster
Dr Line Bay
Prof Madeleine van Oppen
Dr Sven Uthicke
Dr Heidi Luter
Dr James Bell
Students: Mr Jose Montalvo-Proano, Ms Nora Kandler
As part of a long-term mesocosm experiment named 'Evolution 21', we are assessing how current and future climate scenarios (temperature and pCO2) will impact the evolution of reef species. Our research is assessing the inheritance of phenotypic traits that underpin growth and survival including stress tolerance. We quantify a range of traits including host and symbiont gene expression, microbial community composition and function, and host energetics to reveal the mechanisms reef species use to acclimatise and adapt to climate change. This mesocosm experiment aims to closely replicate realistic reef conditions and includes organisms spanning from corals, sponges, foraminifera, seagrasses, urchins to giant clams.
'Evolution21' is using nine mesocosm systems to examine the genetic and non-genetic responses of various reef taxa to 3 climate regimes.
Genetic markers of climate change adaptation in corals
The ability to use quantitative genetic breeding designs allows us to assess the adaptive potential of coral.
AIMS, Oregon State University USA
Dr Line Bay
Ms Holland Elder (NSF GROW Fellow)
Prof Eli Meyer
Persistence of coral species into the future will require adaptation to increased temperatures, with models suggesting that a 1.5°C increase in heat tolerance is required for survival in the immediate future.
Research suggests that the response to selection is determined by the heritability of tolerance traits. Using SeaSim facilities, this team will rear a large number of coral larvae from controlled crosses of parental colonies of the coral species Platygyra daedalea. We will inoculate coral families with cultured Symbiodinium to quantify the roles of parental genetics and symbiosis on heat tolerance. Our analyses will allow us to identify genomic regions under heat selection to develop markers of stress tolerance (i.e., Quantitative Trait Loci or "QTLs"). QTLs can be used to quantify the distribution of tolerant gene variants in coral populations across natural environmental gradients. They can also be used to identify stock for selective breeding and restocking.
Studies of gene function using novel techniques
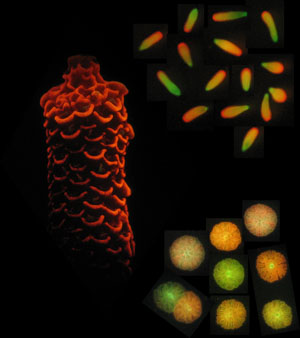
The larvae, juveniles and adults of the coral Acropora millepora are highly fluorescent.
AIMS, University of Texas, Stanford University
Marie Strader
Dr Phil Cleves
Prof Mikhail Matz
Dr Line Bay
In this project, we will test the function of genes with putative roles in coral settlement and metamorphosis. To do this, we will employ multiple methods including RNAi and CRISPR-Cas gene editing using transfection and microinjection. The aim is to knock down expression of six target genes including fluorescent proteins, which are the pigments that make the larvae glow green and red. We will use embryos and larvae of Acropora millepora reared in large batch cultures in SeaSim for our experiments.
We will also measure relevant larval phenotypes in 'treated' and 'untreated' larvae including settlement behaviour, metamorphosis, skeletal development and thermal tolerance. These are traits are key for the survival of young corals. Our work will also reveal specific genes function in corals, with large implications for our understanding the molecular basis for health in these ecologically important species."
Freshly collected egg and sperm bundles from two different coral colonies have different colours. The eggs float to the surface, sperm clouds the water throughout the collection vessel. Pigments and other biochemical compounds determine the colour of eggs and may be linked to the health of the parental colonies. Image: M. Strader
Understanding crown-of-thorns starfish outbreaks
The COTS larval rearing room within the National Sea Simulator.
AIMS, University of Otago, University of Sydney, University of Malaysia
Dr Sven Uthicke
Dr Miles Lamare
Dr Maria Byrne
Dr Aminur Rahman
The Crown-of-Thorns Starfish (COTS) is a natural predator of corals in the Indo-Pacific region, including the Great Barrier Reef (GBR). COTS have been identified, together with cyclones, as a major cause of the 50% decline in coral cover on the GBR observed during the last 27 years. Since the 1960’s, COTS populations have erupted at approximately 15-year intervals with three major outbreaks recorded and a fourth now in progress on the northern GBR. When COTS occur in plague proportions they can reduce the living coral cover on a reef to a few percent. Research continues to reveal more factors playing a role in initiating and maintaining COTS outbreak, which means that the prediction of the effects of climate change on these processes has a high uncertainty.
During the spawning season, we conduct multiple experiments in which we raise COTS larvae until settlement. Most of these experiments test the importance of food (small algae) in the survival of the COTS larvae, but we also test global change-related factors. With help of SeaSim staff, we have developed a flow through larval culture system which allows raising larva under more natural conditions. Larvae cultured in the SeaSim also assist in other research, such as developing eDNA approaches to monitor COTS larvae and adults.